Every time a candle gutters, a diesel engine fires, or a wood-burning stove consumes its fuel, a chemical transformation of extraordinary complexity is taking place. The visible black residue that coats chimney flues, soils ceilings above oil lamps, and darkens the interiors of old furnaces is not simply "burnt material." It is a structured nanomaterial, built molecule by molecule in a fraction of a second, whose chemistry has occupied combustion scientists for the better part of a century.
What Is Soot, Exactly?
Soot is a carbonaceous solid formed as a byproduct of the incomplete combustion of organic matter. It is composed primarily of carbon — typically 60 to 90 per cent by mass in freshly formed particles — but also carries significant quantities of hydrogen, oxygen, nitrogen, sulfur, and a complex array of organic compounds including polycyclic aromatic hydrocarbons (PAHs). The black colour that makes soot so visually distinctive arises from its ability to absorb light across the entire visible spectrum, a consequence of its quasi-graphitic carbon structure and the near-continuum of electronic transitions available to its delocalized electrons.
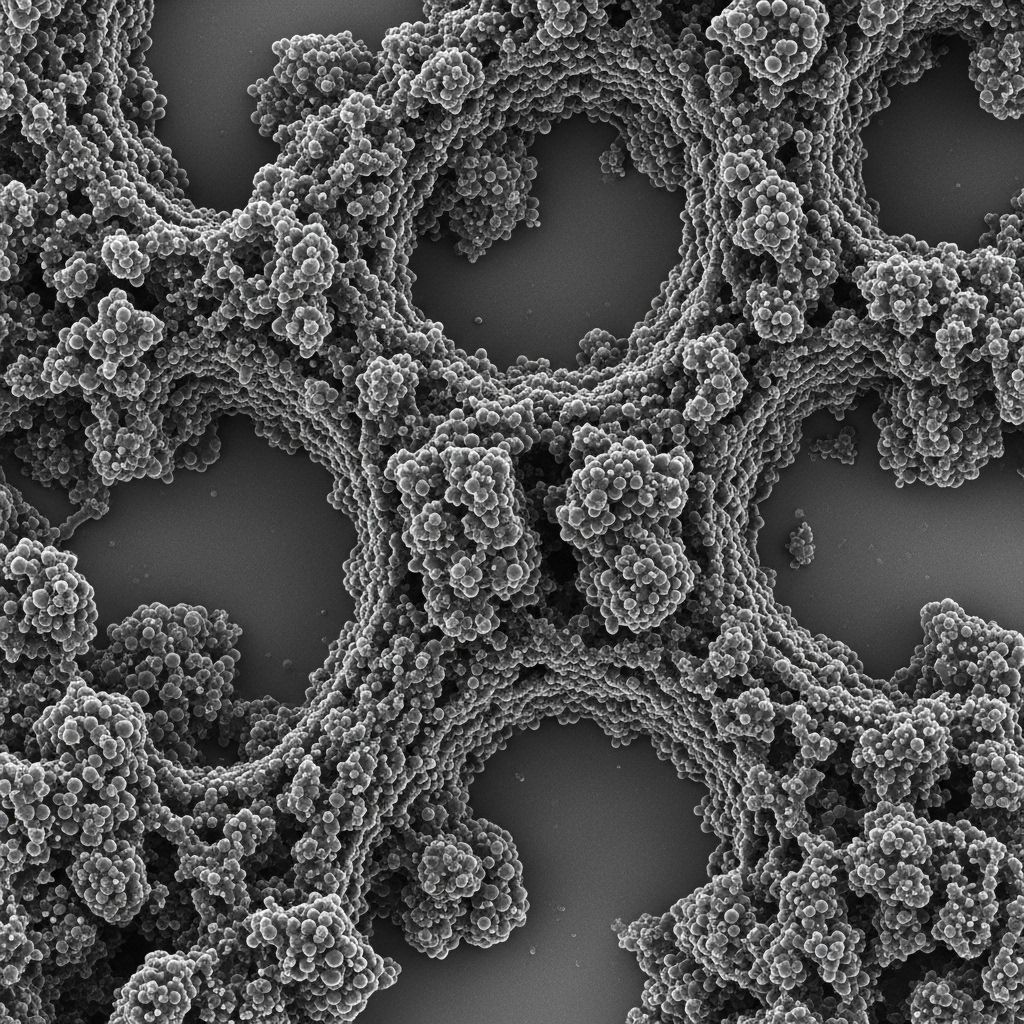
At the nanoscale, soot particles are not homogeneous spheres of carbon. Each primary spherule — the smallest structural unit, typically 10 to 80 nanometres in diameter — has an internal organisation that resembles a crumpled or concentric set of curved graphene-like layers, often described as turbostratic carbon. These spherules then cluster together into chain-like aggregates through van der Waals forces and partial fusion, and these aggregates in turn form larger agglomerates that range from sub-micrometre to several micrometres in effective diameter. It is these agglomerates that we see settling on surfaces as visible soot deposits.
"Soot is among the most structurally complex materials produced by any industrial or natural process — a nano-structured solid whose architecture is written in the physics of turbulent flames."— H. Richter & J.B. Howard, Progress in Energy and Combustion Science, 2000
The Formation Pathway: From Fuel Molecule to Solid Particle
The transformation from a gaseous fuel — say, methane or the long-chain hydrocarbons of diesel — to a solid soot particle proceeds through a sequence of chemical steps that have been elucidated, gradually, over the past seven decades of combustion research.
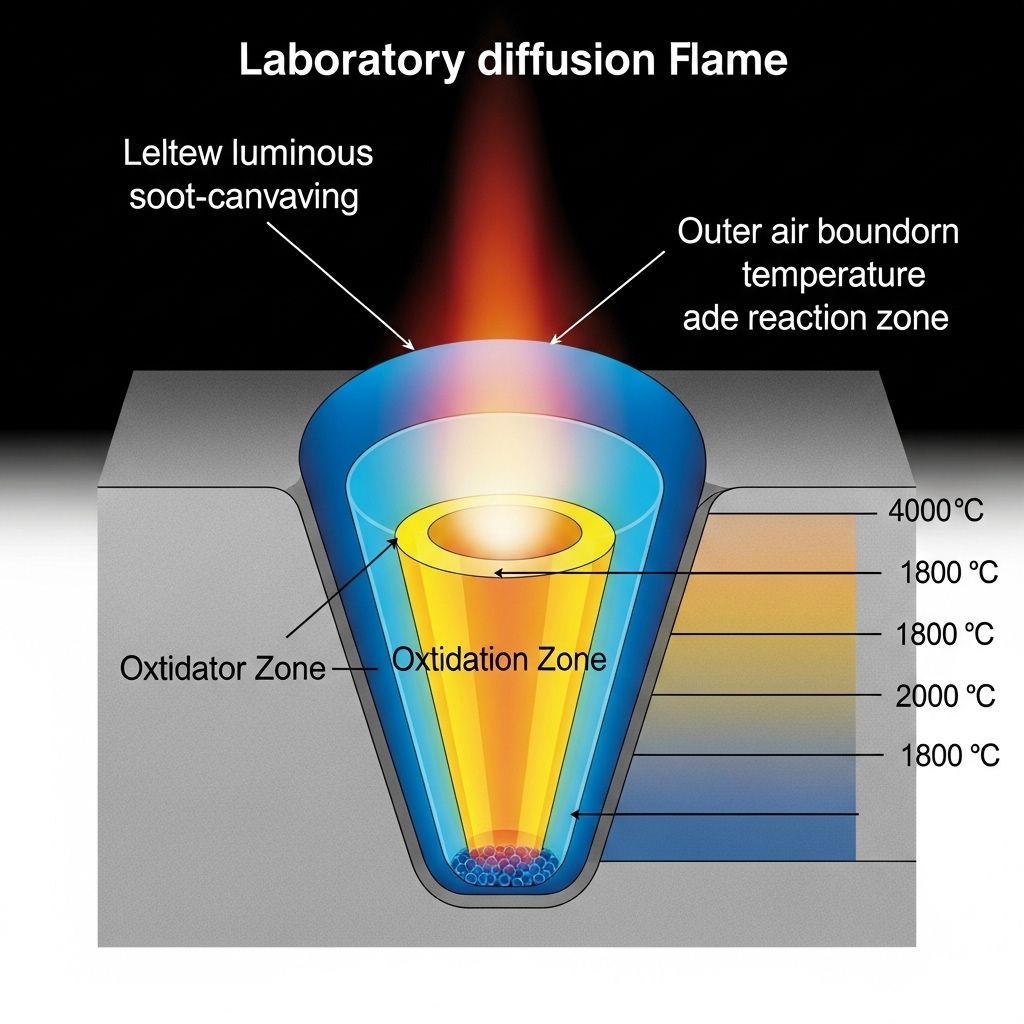
Pyrolysis and radical formation. In the oxygen-deficient inner zone of a diffusion flame, fuel molecules do not combust directly. Instead, they undergo pyrolysis — thermal decomposition in the absence of sufficient oxidant. This produces a variety of smaller reactive fragments: methyl radicals (CH₃), vinyl radicals (C₂H₃), acetylene (C₂H₂), and the propargyl radical (C₃H₃), among others. The propargyl radical deserves special mention: its self-recombination is now understood to be a primary route to the first aromatic ring, benzene (C₆H₆), in combustion systems.
Aromatic ring formation and PAH growth. Once benzene is formed, the flame chemistry takes a decisive turn toward larger structures. Naphthalene, pyrene, coronene — the polycyclic aromatic hydrocarbons are built sequentially through the hydrogen-abstraction acetylene-addition (HACA) mechanism, first proposed by Frenklach and Wang in 1991. In the HACA cycle, a hydrogen atom is abstracted from an existing aromatic molecule, creating a reactive radical site; an acetylene molecule then adds to this site, and after further rearrangements, the ring count increases by one. Each cycle adds two carbon atoms and extends the planar aromatic backbone. Repeated thousands of times per molecule per second in the flame's soot-forming zone, this mechanism produces PAH molecules of increasing molecular weight and decreasing hydrogen-to-carbon ratio.
Nucleation: the gas-to-particle transition. At a molecular weight of roughly 500 to 1000 daltons, PAH molecules begin to reach a critical size at which the dispersion forces between molecules become strong enough to allow spontaneous aggregation. This gas-to-particle transition — nucleation — represents the birth of the first solid-phase soot particles. The precise mechanism remains an area of active research; dimerisation of large PAH molecules, ionic pathways involving polycyclic aromatic hydrocarbon cations, and resonantly stabilised free radical pathways have all been proposed as contributing routes.
Surface growth and coagulation. The nascent nuclei — only 1 to 2 nm in diameter — rapidly acquire mass through two parallel processes. Surface growth involves the addition of gaseous species (acetylene, PAHs, and other hydrocarbons) to the surface of existing particles; this is the dominant mass-addition pathway and is responsible for the bulk of the carbon in a mature soot particle. Simultaneously, coagulation — the collision and sticking of particles — converts many small particles into fewer larger ones, determining the final particle size distribution.
Oxidation. In the outer, oxygen-rich regions of the flame, soot particles encounter OH and O radicals. Surface oxidation by OH is particularly effective at stripping carbon from the particle surface and is the primary mechanism by which flames can be made to burn relatively cleanly. In well-designed combustion systems, most of the soot formed in the fuel-rich core is oxidised before it can escape as emissions. When combustion is poorly designed — or when fuel-to-air ratios are poorly controlled — this oxidation step is incomplete, and soot is emitted.
The Chemical Composition of Collected Soot
When soot deposits are collected and analysed — from a chimney, an engine exhaust pipe, or a laboratory sampling probe — their chemical composition reflects the entire history of their formation and, crucially, what happened to them after the flame. Freshly formed soot from a laboratory burner may contain 80–90% carbon by mass. Soot sampled from a diesel exhaust system or extracted from an old furnace flue can be considerably more complex, owing to the condensation of semi-volatile organic compounds (SOCs), lubricating oil residues, and sulfate salts onto the particle surfaces as they cool.
Of particular concern are the PAHs themselves, some of which are potent carcinogens. Benzo[a]pyrene — a five-ring PAH with a molecular weight of 252 g/mol — is classified as a Group 1 human carcinogen by the International Agency for Research on Cancer (IARC) and serves as a marker compound for PAH contamination in environmental monitoring. Other regulated compounds include fluoranthene, chrysene, and benzo[b]fluoranthene. These species are present both within the soot matrix and adsorbed onto particle surfaces, from which they can desorb and enter the gas phase or be directly ingested when fine particles are inhaled into the deep lung.
Key Terms: Soot Chemistry Glossary
- HACA Mechanism
- Hydrogen Abstraction – C₂H₂ Addition: the dominant pathway for PAH growth in flames, adding two carbon atoms per cycle via acetylene incorporation.
- Primary Soot Particle (Spherule)
- The smallest discrete structural unit of a soot aggregate, typically 10–80 nm in diameter with a turbostratic graphitic internal structure.
- Polycyclic Aromatic Hydrocarbon (PAH)
- A class of organic compounds containing multiple fused aromatic (benzene) rings. Molecular precursors to soot and significant environmental contaminants.
- Turbostratic Carbon
- A form of carbon in which graphene-like sheets are stacked with random rotational and translational offsets — less ordered than graphite but more ordered than amorphous carbon.
- Adiabatic Flame Temperature
- The theoretical maximum temperature attained by combustion products when no heat is lost to the surroundings. For methane–air at stoichiometry: approximately 1950°C.
Soot in the Context of Real Furnaces and Stoves
The chemistry described above plays out wherever fuel is burned — in car engines, power station boilers, biomass stoves, and domestic gas furnaces alike. In a properly maintained and well-tuned residential gas boiler, the combustion process is near-complete: the air-to-fuel ratio is carefully controlled to provide a slight excess of oxygen, temperatures in the primary combustion zone exceed 1100°C, and the flue gas residence time is long enough for soot oxidation to proceed effectively. Soot emissions from a modern condensing gas boiler are typically measured in single-digit milligrams per cubic metre of flue gas — essentially negligible.
The situation is different for older, poorly maintained, or improperly operated systems. A wood-burning stove running on wet (high moisture content) timber, an oil-fired boiler with a clogged or misadjusted burner nozzle, or a gas furnace with a dirty heat exchanger will all operate with sub-optimal combustion conditions. The result is visible as soot deposits in the flue: black, oily, tar-like accumulations that grow on flue-liner surfaces, creosote-rich in the case of wood smoke, sulfate-laden in the case of sulfur-containing fuels.
These deposits are more than an aesthetic problem. Thick soot layers in a chimney reduce the effective draught (by narrowing the flue cross-section), insulate the flue gases (reducing buoyancy and further weakening draught), and — in extreme cases — create a fire hazard: chimney fires, in which accumulated creosote deposits ignite, can generate flue gas temperatures exceeding 1000°C in residential chimneys designed to operate below 400°C.
Physical Properties Relevant to Engineering
For the thermal engineer, soot is not merely a contamination problem — it is also a heat transfer medium. The emissivity of soot-laden flames is substantially higher than that of transparent flames: a luminous, soot-containing diffusion flame can radiate 20–40 times more thermal energy per unit volume than a non-luminous premixed flame at the same temperature. This property is exploited in industrial furnaces where radiant heat transfer to the workpiece is desired. In steel reheating furnaces, glass-melting tanks, and cement kilns, the presence of luminous flames — and therefore soot — is not a failure but a design choice.
The specific surface area of soot is enormous: values of 50–200 m²/g are commonly reported, depending on the degree of aggregation and the surface chemistry. This large surface area makes soot a highly effective carrier for adsorbed chemical species, an important consideration in both atmospheric chemistry (where soot aerosols transport PAHs and heavy metals from combustion sources to remote environments) and in industrial carbon chemistry (where controlled surface area is a key specification for engineered carbon black products).
Atmospheric and Environmental Significance
Black carbon aerosol — essentially atmospheric soot — is recognised by climate scientists as the second-largest contributor to anthropogenic climate warming after carbon dioxide, with a global mean forcing estimated at approximately 0.88 W/m² (Bond et al., 2013, Journal of Geophysical Research). This forcing operates through multiple mechanisms: direct absorption of solar radiation; semi-direct effects whereby the heated black carbon layer alters cloud formation; and indirect effects through the reduction of surface albedo when black carbon deposits on snow and ice.
In the Netherlands, black carbon emissions have declined substantially since the 1970s as coal combustion has been phased out of domestic heating, industrial processes have been regulated under EU industrial emissions legislation, and vehicle emissions standards have tightened. Nevertheless, residential wood burning — which has grown as a perceived "sustainable" heating option — has emerged as a significant local source of both black carbon and PAH emissions, particularly in winter when meteorological conditions suppress atmospheric mixing.
Conclusion: A Material That Writes Its Own History
Soot occupies an unusual position in science: it is simultaneously a nuisance, a hazard, a climate forcer, a raw material, and — in the hands of an electron microscopist — one of the most structurally fascinating materials in the natural world. Its chemistry encodes the conditions under which it was formed: the fuel type, the temperature distribution, the air-fuel ratio, the age of the deposit. A combustion scientist examining a soot sample from a centuries-old Dutch furnace chimney can, in principle, read that deposit as a record of the fuels burned and the conditions prevailing in the stove — a chemical archive of thermal history.
Understanding soot formation is not academic indulgence. It underpins the design of cleaner engines, more efficient industrial furnaces, better air quality regulation, and more accurate climate models. It also helps explain why your chimney sweep insists on annual cleaning — and why, when it is neglected, the consequences can be severe.
Key References
- Frenklach, M. & Wang, H. (1991). Detailed modeling of soot particle nucleation and growth. Symposium (International) on Combustion, 23(1), 1559–1566.
- Richter, H. & Howard, J.B. (2000). Formation of polycyclic aromatic hydrocarbons and their growth to soot — a review of chemical reaction pathways. Progress in Energy and Combustion Science, 26(4–6), 565–608.
- Bond, T.C. et al. (2013). Bounding the role of black carbon in the climate system: A scientific assessment. Journal of Geophysical Research: Atmospheres, 118(11), 5380–5552.
- IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Volume 92: Some Non-heterocyclic Polycyclic Aromatic Hydrocarbons and Some Related Exposures. Lyon: IARC, 2010.
- Williams, A., Jones, J.M., Ma, L. & Pourkashanian, M. (2012). Pollutants from the combustion of solid biomass fuels. Progress in Energy and Combustion Science, 38(2), 113–137.