Ask a chemist to describe soot and carbon black, and they will likely reach for the same vocabulary — turbostratic carbon, polycyclic aromatic hydrocarbons, primary spherule diameter, BET surface area. The two materials share a common chemical family, a common colour, and common manufacturing ancestors. Yet the rubber tyres on a Boeing 777, the ink in a laser printer, and the UV-resistant coating on a garden hose all depend on manufactured carbon black — a material so precisely engineered that its particle diameter is specified to within 5 nanometres and its impurity profile to within 0.1%. Combustion soot, by contrast, is classified by the European Chemicals Agency under the SVHC (Substance of Very High Concern) framework and is subject to occupational exposure limits because of its carcinogenic PAH content.
A Brief History of Carbon Black as an Industrial Material
The use of black carbon as a colouring agent is ancient: cave paintings at Lascaux (circa 17,000 BCE) use charcoal and lamp black — crude carbon black made from incomplete combustion of animal fat in clay lamps — as the dominant black pigment. Chinese ink sticks dating to the 3rd century BCE are formulated from pine soot and glue. In ancient Egypt, kohl eye-liner was partly composed of carbonaceous particles.
The industrial story of carbon black, however, begins in the early 20th century with the discovery that carbon black dramatically improves the mechanical properties of natural rubber. In 1906, Sidney Charles Mote at the Silver Town rubber works in London observed that carbon black incorporated into vulcanised natural rubber increased its tensile strength by a factor of ten compared to unloaded rubber. The mechanism — interfacial bonding between carbon black particles and the rubber polymer chains, reinforced by the enormous surface area of the particles — was not understood for decades, but the empirical observation was immediately applied: by 1915, carbon black was being produced industrially in large quantities for rubber reinforcement, and by 1930 it was present in virtually every pneumatic tyre manufactured in Europe and North America.
The furnace black process — still responsible for more than 95% of global carbon black production today — was developed in the 1940s. In this process, a liquid hydrocarbon feedstock (typically a heavy aromatic oil derived from crude oil refining or coal tar distillation) is partially combusted in a refractory-lined reactor. The conditions — feedstock injection rate, reactor temperature (typically 1200–1800°C), residence time, and quench timing — are precisely controlled to produce a carbon black with the desired particle size, structure (degree of aggregation), and surface chemistry. By varying these parameters, manufacturers can produce grades ranging from N110 (primary particle diameter 11–19 nm, used in high-performance tyres) to N990 (primary particle diameter 285–500 nm, used as a filler in seals and gaskets).
"Carbon black is perhaps the only material where the manufacturer specifies not just composition but the three-dimensional architecture of the particle itself — its size, its branching structure, its surface texture — with the precision of a pharmaceutical product."— R. Donnet, R.C. Bansal & M.J. Wang, Carbon Black: Science and Technology, 2nd edn., 1993
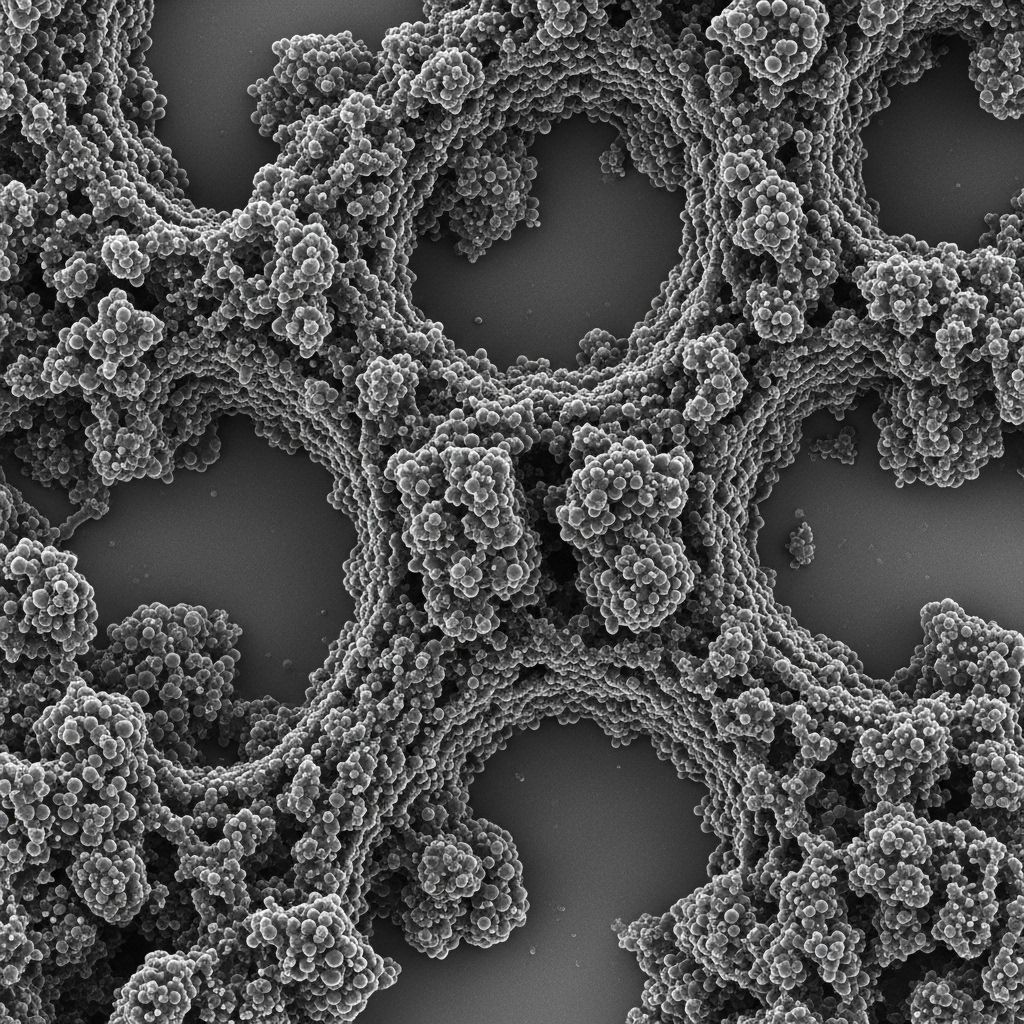
Molecular Architecture: What the Electron Microscope Reveals
Both industrial carbon black and combustion soot are built from the same basic structural unit: the primary spherule, a roughly spherical particle whose interior consists of short-range-ordered graphene-like layers (typically 3–8 layers per stack, each stack 1–3 nm across) arranged in a turbostratic, onion-like configuration. At this scale, the materials look superficially similar in a transmission electron microscope. The differences emerge at several levels.
Particle size distribution. Industrial carbon black grades are manufactured to a narrow, specified primary particle size distribution: the standard deviation in primary particle diameter for a typical furnace black is ±10–15% of the mean. Combustion soot has a much wider distribution, reflecting the spatially and temporally heterogeneous conditions in a real flame: particles formed in different zones of the flame have experienced different temperatures and residence times, yielding a broad size distribution.
Chemical purity. High-quality furnace carbon black contains 95–99% elemental carbon, with tightly controlled residual hydrogen (0.3–0.5%), oxygen (0.1–1.0%), and ash (0.1–0.5%) contents. Combustion soot contains 40–90% carbon (highly variable depending on fuel and combustion conditions), with substantial quantities of hydrogen (3–8%), oxygen (3–10%), sulfur (up to 5% in oil or coal combustion), nitrogen compounds, heavy metals, and PAHs. The PAH content is particularly significant: freshly collected diesel exhaust soot can contain 1–5% PAHs by mass, compared to less than 0.1% in commercial carbon black.
Surface chemistry. Carbon black manufacturers control surface functional groups (primarily carboxyl, lactol, quinone, and phenol groups on the particle surface) to tailor the material's interaction with polymers and solvents. Combustion soot surfaces are loaded with a heterogeneous mixture of adsorbed organic compounds, including semi-volatile PAHs, nitro-PAHs, and oxygenated PAHs (oxy-PAHs) — compounds whose surface binding and desorption characteristics are poorly controlled and which represent the primary biological hazard of soot particle inhalation.
The Surface Area Question: Why Nanoscale Structure Matters
The specific surface area of carbon black — typically measured by the BET (Brunauer–Emmett–Teller) nitrogen adsorption method and expressed in m²/g — is the single most important parameter determining its reinforcing effectiveness in rubber and its performance in other applications. BET surface areas for commercial grades range from approximately 9 m²/g (N990 thermal black) to over 1400 m²/g (certain speciality grades used in battery electrodes and capacitors).
Combustion soot has highly variable BET surface areas depending on its source and collection conditions: freshly formed soot particles in the flame may have surface areas of 100–200 m²/g, but aged, agglomerated, and surface-coated soot from a chimney deposit may have an apparent BET surface area of only 10–50 m²/g due to particle fusion and pore blockage by condensed organics.
The high surface area of carbon black is not merely a curiosity of particle physics — it is the mechanistic basis for its utility. In a rubber compound, carbon black particles bond to the polymer chains across their enormous surface, creating a three-dimensional network of polymer-particle-polymer bridges that dramatically increases the elastic modulus, tear strength, and abrasion resistance of the compound. The rubber tyres that consume approximately 70% of global carbon black production depend entirely on this surface area effect: a tyre without carbon black reinforcement would have less than one-tenth the service life.

Classification, Regulation, and the PAH Hazard
The regulatory treatment of carbon black and combustion soot reflects their fundamentally different risk profiles. Commercial carbon black used in rubber and plastic compounding is not classified as a carcinogen in the EU Classification, Labelling and Packaging (CLP) Regulation, provided its benzo[a]pyrene content is below 1 mg/kg. The International Agency for Research on Cancer (IARC) classifies carbon black as a Group 2B possible human carcinogen — based on limited evidence in animal studies and inadequate evidence in humans — with the caveat that the evidence relates primarily to occupational inhalation exposure at high dust concentrations rather than to the material's chemical properties per se.
Combustion soot occupies a very different regulatory category. Diesel engine exhaust — which is predominantly a soot aerosol with adsorbed organic compounds — was reclassified by IARC in 2012 from Group 2A (probably carcinogenic) to Group 1 (definitely carcinogenic to humans), based on epidemiological evidence from cohort studies of underground miners, truck drivers, and railway workers. The carcinogenic activity of diesel exhaust soot is attributed primarily to its high PAH content, particularly benzo[a]pyrene and other nitro-PAHs, rather than to the carbon matrix itself.
This regulatory distinction has practical implications for industries handling both materials. Carbon black is subject to a workplace exposure limit (WEL) of 3.5 mg/m³ (inhalable fraction) and 1.5 mg/m³ (respirable fraction) in the Netherlands, as set by the SER (Social and Economic Council) occupational exposure standards. Soot from combustion processes is regulated as a carcinogen under the Dutch Arbeidsomstandighedenbesluit (Working Conditions Decree), requiring engineering controls to minimise exposure to as low as reasonably practicable.
Emerging Applications: When Soot Becomes Useful
The sharp functional distinction between manufactured carbon black (useful, controlled, commercially valuable) and combustion soot (a waste product and health hazard) is beginning to blur at the frontiers of materials research. Researchers at the University of Groningen and TU Eindhoven have explored the direct collection of carbon nanoparticles from controlled combustion processes as a low-cost alternative to conventional carbon black manufacturing — potentially reducing the energy and hydrocarbon feedstock cost of carbon black production by 30–40%.
Soot from biomass combustion, when collected under controlled conditions and subjected to post-processing to remove PAH contamination (typically by washing with organic solvents and mild thermal treatment), has been investigated as a soil amendment (biochar) with potentially beneficial effects on soil carbon sequestration and water retention. This application — essentially the controlled deployment of combustion carbon residues in agricultural contexts — represents a striking conceptual inversion of the usual treatment of soot as a waste problem.
The molecular science that distinguishes carbon black from soot — the control of particle size, surface chemistry, structural order, and chemical purity — is also increasingly relevant to the emerging field of nanocarbon materials, where fullerenes, carbon nanotubes, and graphene occupy a continuum of structural perfection above the "engineering carbon" range occupied by carbon black. Understanding where soot ends and engineered nanocarbon begins requires exactly the kind of molecular-level analysis that this review has attempted to lay out.
Key References
- Donnet, J.B., Bansal, R.C. & Wang, M.J. (eds.) (1993). Carbon Black: Science and Technology, 2nd edn. Marcel Dekker, New York.
- IARC (2010). Carbon Black, Titanium Dioxide, and Talc. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Volume 93. IARC, Lyon.
- IARC (2012). Diesel and Gasoline Engine Exhausts and Some Nitroarenes. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Volume 105. IARC, Lyon.
- Fonseca Couto, C., Lange, L.C. & Santos Amaral, M.C. (2018). A critical review on membrane separation processes applied to remove pharmaceutically active compounds from water and wastewater. Journal of Water Process Engineering, 26, 156–175.
- Olfert, J. & Rogak, S. (2019). Universal relations between soot effective density and primary particle size for common combustion sources. Aerosol Science and Technology, 53(5), 485–492.